SSRI Co., Ltd.
NEWS
SSRI Co., Ltd.
Rare Disease Day 2026
- Toward Eliminating "Drug Loss":
Data on Disparities in Access to Treatment and the Future of Collaborative Innovation -

The last day of February is a day to shine a light on rare and intractable diseases.
The last day of February is a day to shine a light on rare and intractable diseases.
Every year, the final day of February marks Rare Disease Day (hereafter RDD).
The theme in Japan for 2026 is: "Together, we live. Together, we create. At times, we smile. RDD: Readiness, Dialogue & Discovery."
In the field of rare and intractable diseases, patients and their families often face isolation. To overcome this reality, patients, healthcare professionals, government, and society as a whole must share a spirit of Readiness, Dialogue, and Discovery, working together to build new frameworks for treatment and support.
The Pressing Reality of Rare and Intractable Diseases and "Drug Loss"
While more than 7,000 rare diseases exist globally, affecting an estimated 300 million patients, the difficulty of ensuring commercial viability in drug development creates structural challenges that often slow progress. In Japan, 348 diseases (as of April 2025) are designated as intractable diseases, but the small number of patients makes it difficult to conduct clinical trials, so drug development tends to lag behind that for more common diseases.
In recent years, "drug loss" has become a pressing issue. The term refers to a loss of access to treatment opportunities, in which drugs already approved overseas have not even reached the development-planning stage in Japan. According to a Ministry of Health, Labour and Welfare survey (as of March 2023), of 86 drugs approved in Europe and the United States but not yet initiated for development in Japan, approximately half (40) were treatments for rare diseases. Particularly in rapidly progressing pediatric conditions, even a delay of a few months can directly lead to irreversible disability or impact life expectancy, making this a challenge that cannot be ignored from a humanitarian standpoint.Data Analysis: Highlighting the Japan–U.S. Gap in Pediatric Treatment
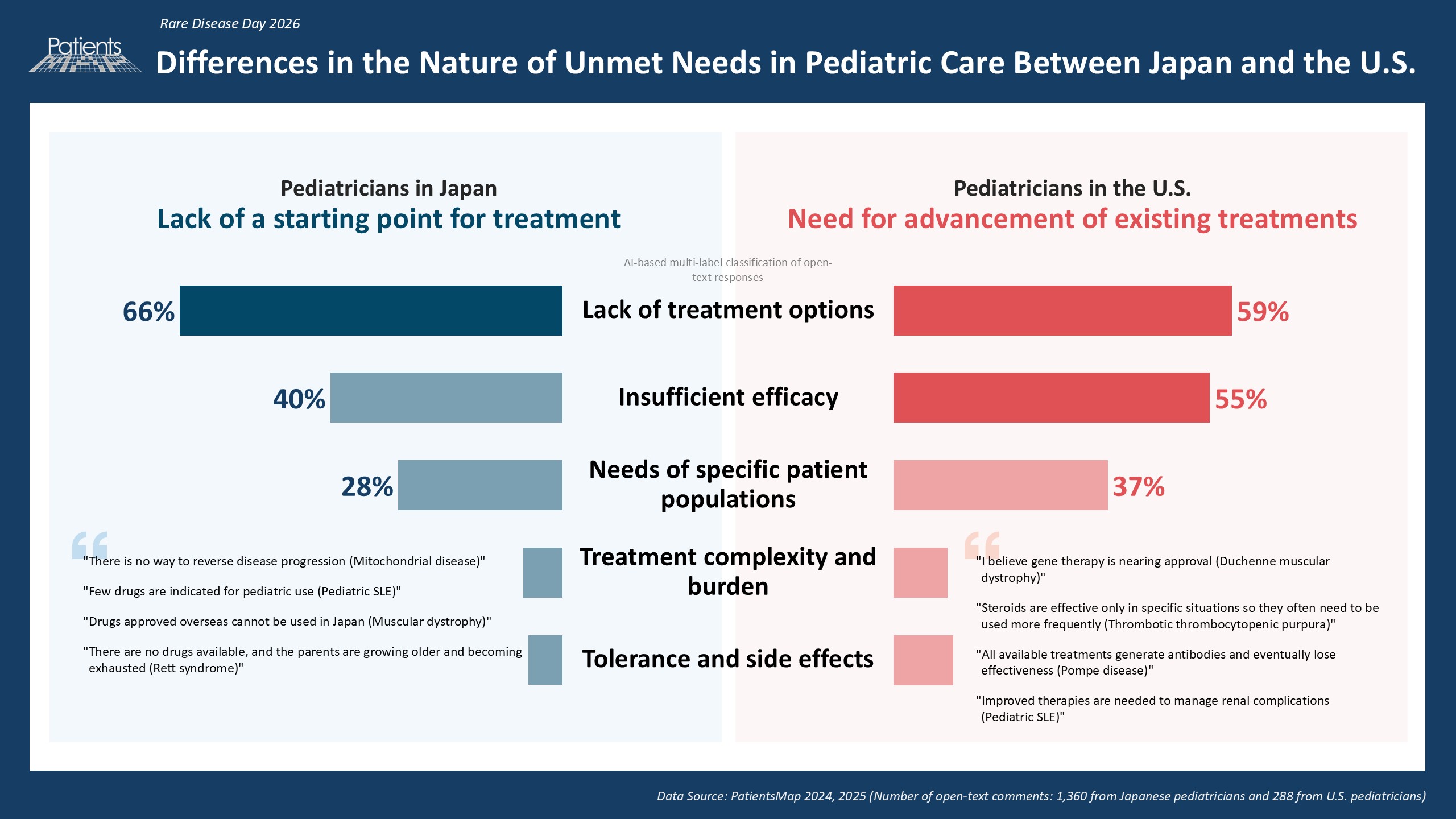
Against this background, we used the physician research database PatientsMap to compare and analyze unmet needs faced by physicians in pediatrics, where timely treatment is critical, examining differences between Japan and the U.S.
Analysis Methodology:This analysis covered 142 rare and intractable diseases included in both the Japan and U.S. editions of PatientsMap. For physicians who treat patients with these conditions and indicated that they "desire new drugs," we utilized the extensive open-text responses they provided on their reasons for desiring new drugs and on their dissatisfaction or issues with current treatments. These text data were analyzed using AI (natural language processing)–based multi-label classification, enabling comprehensive, quantitative aggregation of multiple needs—such as lack of treatment options and insufficient efficacy—and visualization of the challenges faced in clinical settings in Japan and the United States.
(AI-based multi-label classification of open-text responses)
- Japan: Not Even at the "Starting Line" of Treatment
The analysis revealed that 66% of pediatricians in Japan report a lack of treatment options. This figure exceeds that of pediatricians in the United States (59%). Meanwhile, in the U.S., dissatisfaction regarding insufficient efficacy (55%) was higher than in Japan (40%), pointing to a stronger demand for improvement in the quality of existing treatments. In contrast, the findings suggest that the most urgent priority in Japan is to secure fundamental treatment options in the first place.Comments from Pediatricians in Japan (excerpts):
"There is no way to reverse disease progression (Mitochondrial disease)"
"Few drugs are indicated for pediatric use (Pediatric SLE)"
"Drugs approved overseas cannot be used in Japan (Muscular dystrophy)"
"There are no drugs available, and the parents are growing older and becoming exhausted (Rett syndrome)"Comments from Pediatricians in the United States (excerpts):
"I believe gene therapy is nearing approval (Duchenne muscular dystrophy)"
"Steroids are effective only in specific situations so they often need to be used more frequently (Thrombotic thrombocytopenic purpura)"
"All available treatments generate antibodies and eventually lose effectiveness (Pompe disease)"
"Improved therapies are needed to manage renal complications (Pediatric SLE)" - Frontline Perspectives: Contrasting Burdens in Japan and the U.S.
Based on an analysis of open‑text responses, a comparison of the nature of the challenges faced by pediatricians in Japan and the United States reveals a clear disparity.
Japanese physicians speak of a sense of helplessness stemming from the lack of available treatment options, whereas in the United States the discussion centers on more advanced‑stage medical challenges—such as overcoming side effects and achieving a cure—premised on the existence of established treatment choices.Pediatricians in Japan Pediatricians in the U.S. Primary burdens Lack of a starting point for treatment
"There is not a single drug available"
"All we can do is watch the disease progress."Need for advancement of existing treatments
"Drugs are available, but their side effects are severe"
"Current drugs do not lead to a complete cure"Keywords "Gap" "Helplessness" "Starting point" "Quality" "Avoidance of side effects" "Complete cure" Future Aim To reach the starting point of treatment as a first step Shifting away from steroids toward a complete cure through gene therapy
Hope for the Future: Government Support to Accelerate in FY2026
In order to overcome this stagnation, comprehensive reforms to Japan's drug development landscape will get underway in FY2026.
The Ministry of Health, Labour and Welfare plans to expand the Program to Secure Access to Unapproved and Off‑Label Drugs, which offers direct government funding for clinical trials focused on ultra-rare diseases with patient populations of only a few dozen. Furthermore, by strengthening the pediatric drug development network, an efficient trial framework is being formed around key institutions including the National Center for Child Health and Development.
In Closing
On Rare Disease Day 2026, we renew our call to society to re-examine the issue of "drug loss."
Numbers alone do not tell the full story. Behind the data lie patients awaiting treatment and physicians hoping they can help provide it. We seek to build a society in which every patient receives an accurate diagnosis and has access to the treatment they need. Together, we aim to build a future where no one has to forgo treatment simply because they happen to live in Japan—a future where we can "smile together."
Preparation of This Article
This article was prepared in alignment with the objectives of RDD 2026 and is based on physician survey data from Japan and the United States (PatientsMap) as well as publicly available materials from the Ministry of Health, Labour and Welfare.
Related Links
Official RDD Japan Website:https://rddjapan.info/2026/
Analysis data:https://research.ssri.co.jp/pmap/
Inquiries
SSRI Co., Ltd.
patientsmap@ssri.com